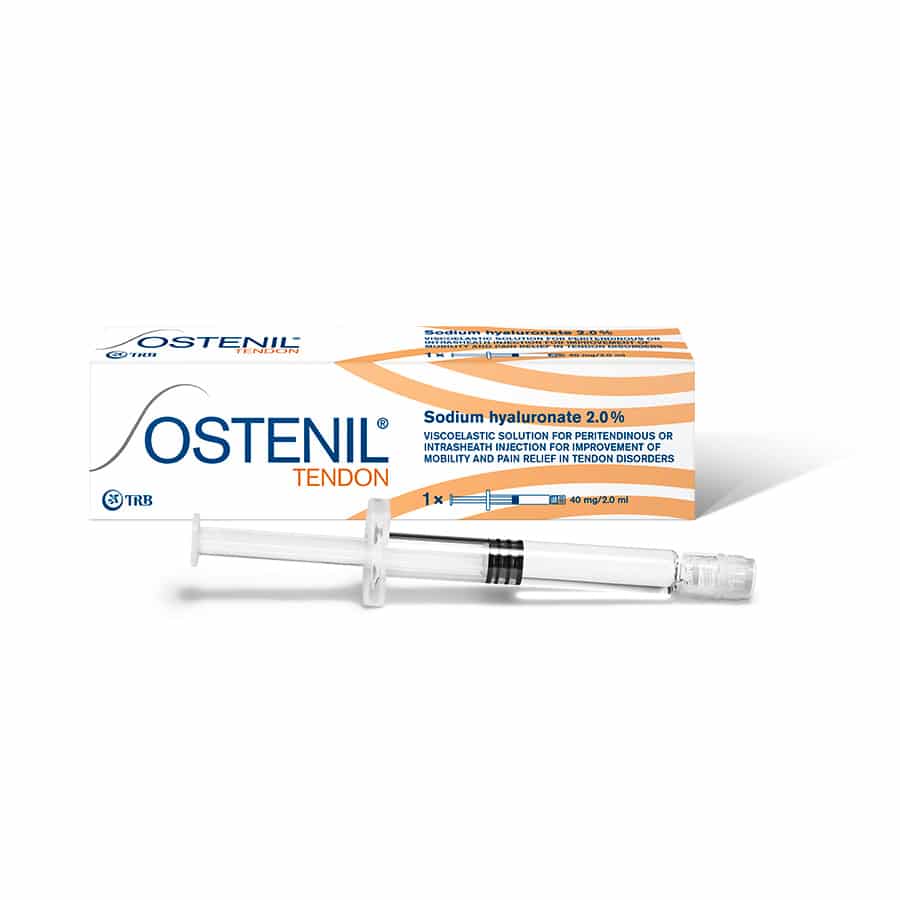
OSTENIL® TENDON
Pre-filled syringe 2 ml
OSTENIL® TENDON pre-filled syringe is formulated to relieve pain and improve mobility in the case of tendon disorders. Effective, safe and very well-tolerated!
OSTENIL® TENDON is injected by the doctor into the tendon sheath of affected tendon. If tendon sheath is absent, a peritendinous injection can be done. In both cases, OSTENIL® TENDON will spread throughout the tendon. OSTENIL® TENDON has an ideal compatibility as it contains hyaluronic acid that matches the natural hyaluronic acid present in human body.
- 2% hyaluronic acid obtained by biofermentation
- 0.5% mannitol
- 2 injections at weekly interval
- Suitable for all tendons
Registered under Act 737
MDA Reg. No. GD64411550418